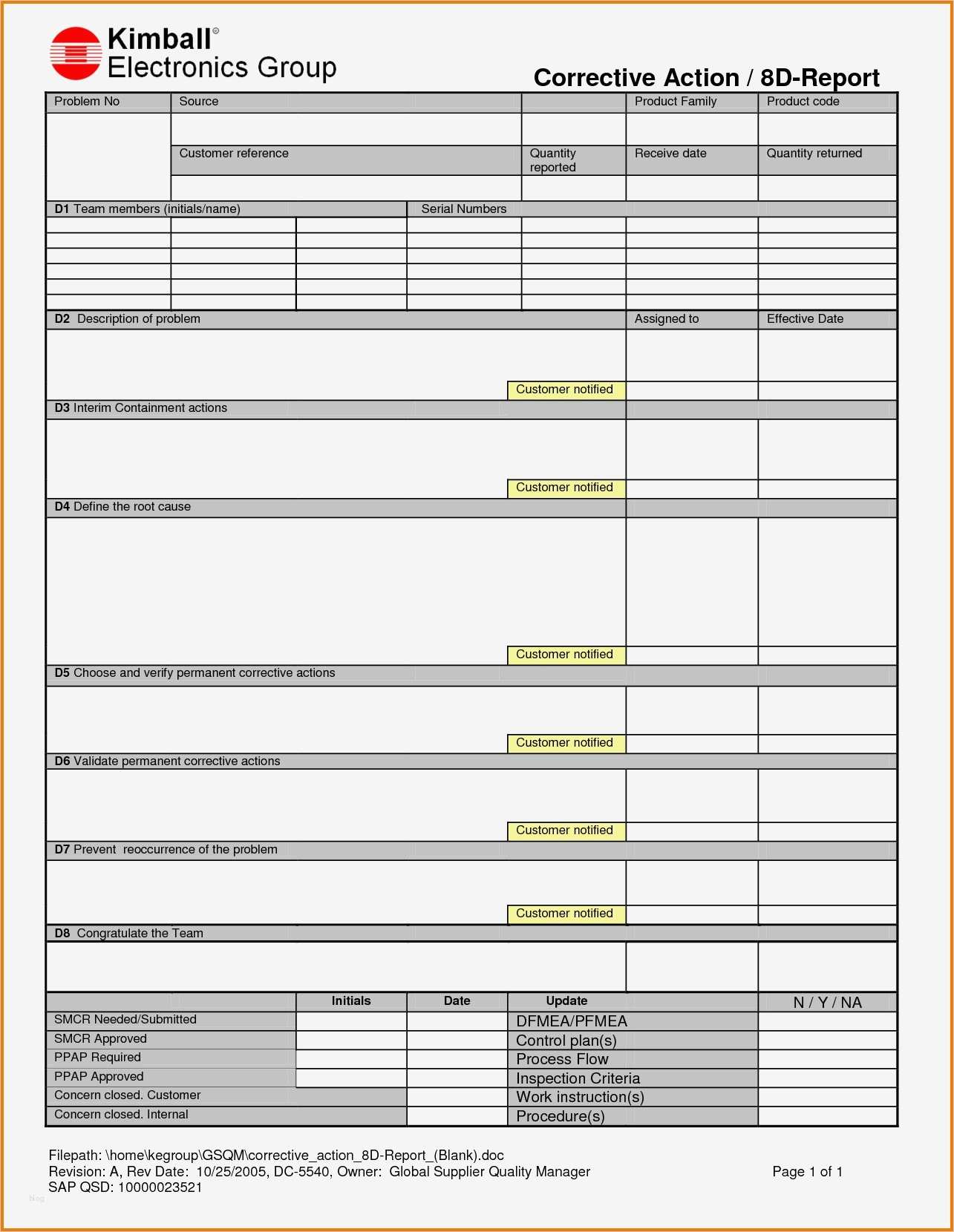
There should be nothing extra in the procedure, and nothing left out. As an author, you should use an outline format and match your CAPA form exactly. An author may abbreviate a step or skip it altogether. Procedures are often unclear because the author is an expert with more experience than the intended audience. Medical Device Academy’s updated CAPA procedure is only six pages and the CAPA form is four pages. Procedures are often unclear because the author is more familiar with the process than the intended audience for the procedure. Make sure your CAPA procedure is clear and concise This client purchased their CAPA procedure from another consultant, changed the title, and had everyone in the company “read and understand” the procedure for training. The procedure and the process MUST match. The procedure was 100% compliant with FDA regulations, but the procedure did not match how the company performed the process. This example is a minor nonconformity, but the reason why this client was not following their procedure is more interesting. Despite this requirement in their procedure, the client was indicating that CAPA plans were due within 30 calendar days on their CAPA form. Specifically, the procedure indicated that all CAPA plans must be written within seven calendar days of initiating the CAPA. 
CAUTION: Read the story in the next few paragraphs before you implement any purchased procedureĭuring a recent internal audit, I noticed that the client was not meeting one of the requirements of their CAPA procedure.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
